When you choose a peptide, the most important parameter determining its quality is not the price, the packaging, or the supplier’s brand. It is a number: the percentage purity determined by HPLC. This seemingly simple value is the result of a sophisticated analysis that reveals precisely what is actually inside the vial you are holding.
Think of peptides like retatrutide — which on this blog we call TRIPLE-G for its three G’s (GLP-1, GIP, Glucagon): it is a complex molecule, with 39 amino acids. Every step of its synthesis can introduce impurities. That is why purity matters so much.
In this article we take a deep dive into everything you need to know about HPLC applied to peptides: how it works, how to interpret the results, why the difference between 95% and 99%+ is far from trivial, and how to verify supplier purity claims.
What Is HPLC?
Definition and Principle
HPLC (High-Performance Liquid Chromatography) is an analytical technique that separates, identifies, and quantifies the components of a liquid mixture. In simple terms, it is the best way we have to measure how pure a peptide is. It is the gold standard worldwide.
The fundamental principle is straightforward: a mixture of compounds is carried through a chromatographic column by a solvent (the so-called “mobile phase”). The different components of the mixture interact differently with the column’s packing material (the “stationary phase”) and are therefore separated based on their physicochemical properties. Components that interact weakly with the stationary phase elute (exit the column) first; those that interact more strongly elute later.
Components of an HPLC System
A typical HPLC system comprises:
- Solvent reservoir: holds the mobile phase (water/acetonitrile mixtures with trifluoroacetic acid are the most common for peptides)
- High-pressure pump: drives the mobile phase through the column at pressures of 50–400 bar
- Injector: introduces the sample (the peptide to be analyzed) into the solvent flow
- Chromatographic column: the heart of the system, typically a C18 reversed-phase column 100–250 mm long with a 4.6 mm internal diameter
- UV detector: measures the ultraviolet light absorption of compounds eluting from the column (peptides typically absorb at 214 nm or 220 nm)
- Data acquisition system: software that generates the chromatogram
Reversed-Phase HPLC (RP-HPLC) for Peptides
For peptide analysis, the most widely used mode is reversed-phase (RP-HPLC). In this configuration:
- The stationary phase is nonpolar (C18 chains bonded to silica)
- The mobile phase is polar (water to acetonitrile gradient)
Peptides, which have both polar and nonpolar regions, are separated based on their hydrophobicity: more hydrophilic peptides elute first, while more hydrophobic ones elute later. This technique is excellent for separating the target peptide from synthesis impurities, which often differ by one or a few amino acids.
How to Read a Chromatogram
The Analysis Output
The result of an HPLC analysis is a chromatogram: a graph with retention time (minutes) on the X-axis and UV signal intensity (milliabsorbance units, mAU) on the Y-axis.
Key Elements of a Chromatogram
The main peak: this is the tallest and most prominent peak in the chromatogram, corresponding to the target peptide. Its position on the X-axis is the retention time — a characteristic value that depends on the peptide’s structure and the analytical conditions. For a given HPLC method, the retention time is reproducible and allows compound identification.
Impurity peaks: these are the smaller peaks that appear before, after, or alongside the main peak. Each peak corresponds to a different impurity — a truncated peptide, an oxidized form, an isomer, or a synthesis byproduct.
The baseline: this is the horizontal line of the chromatogram in the absence of eluting compounds. A stable, flat baseline indicates a well-conducted analysis.
Peak area: purity is calculated as:
Purity (%) = (Area of the main peak / Total area of all peaks) x 100
For example, if the main peak has an area of 990 units and the total area of all peaks is 1,000 units, the purity is 99.0%.
How to Evaluate a Quality Chromatogram
A good chromatogram exhibits:
- Symmetrical main peak: an asymmetrical peak (with “tailing” or “fronting”) may indicate chromatographic issues or a mixture of isomers
- Flat baseline: oscillations in the baseline suggest instrumental problems or solvent contamination
- Good resolution: impurity peaks must be clearly separated from the main peak
- Adequate time scale: the gradient must be long enough to separate the components
The Purity Grading System
Not all peptides are created equal. The industry uses a classification system based on HPLC purity:
Purity Grades
| Grade | HPLC Purity | Typical Use | Relative Cost |
|---|---|---|---|
| Crude | <70% | Not suitable for research | Very low |
| Research grade | >95% | Initial screening, preliminary studies | Low |
| High purity | >98% | Standard research, most studies | Medium |
| Premium | >99% | Precise quantitative studies, in vivo | High |
| Ultra-premium | >99.5% | Reference studies, critical dose-response | Very high |
What This Means in Practice
The numerical difference between 95% and 99% may seem modest — only 4 percentage points. But consider what it means in terms of actual content:
Peptide at 95% purity: out of 10 mg of powder, 9.5 mg is the desired peptide and 0.5 mg is impurities. If you prepare a 5 mg/ml solution, each milliliter contains 0.25 mg of unknown impurities.
Peptide at 99% purity: out of 10 mg of powder, 9.9 mg is the desired peptide and only 0.1 mg is impurities — five times less than 95%.
Peptide at 99.5% purity: out of 10 mg of powder, 9.95 mg is the desired peptide and just 0.05 mg is impurities — ten times less than 95%.
Peptide at 99.8% purity: out of 10 mg of powder, 9.98 mg is the desired peptide and only 0.02 mg is impurities — twenty-five times less than 95%.
Why Purity Matters: The Scientific Reasons
Peptides like GLP-1 are biological messengers that your body already produces naturally — for example, GLP-1 is released after every meal to signal satiety. When you use a synthetic version like semaglutide or TRIPLE-G, you want it to be as identical as possible to the natural signal, without chemical “noise.”
1. Reproducibility of Results
The fundamental principle of science is reproducibility: an experiment conducted under the same conditions must produce the same results. Impurities introduce variability into results because:
- The composition and concentration of impurities vary from batch to batch
- Some impurities may have their own biological activity (agonistic or antagonistic)
- The “noise” from impurities obscures the signal from the target peptide
With a peptide at 99.5%+, the variability introduced by impurities is negligible and results are highly reproducible.
2. Dosage Accuracy
In any dose-response study, dosage precision is critical. If a peptide has a purity of 95%, each nominal dose actually contains 5% less active compound than calculated. For studies measuring fine differences between dosages (e.g., 100 mcg vs. 250 mcg vs. 500 mcg), this imprecision can:
- Shift the dose-response curve
- Alter the EC50 determination
- Produce irreproducible data between different settings using peptides of different purity
3. Reduction of Confounding Effects
Impurities are not inert. They can:
- Generate false positives: an impurity with its own biological activity may produce an effect erroneously attributed to the target peptide
- Cause toxicity: synthesis byproducts (residual coupling agents, solvents) can be cytotoxic
- Interfere with assays: fluorescent impurities or those with UV absorption can interfere with spectrophotometric or immunological assays
4. Lower Effective Dose Required
A practical corollary of high purity is that lower nominal doses are needed to achieve the same biological effect. A peptide at 99.8% requires approximately 5% less material than one at 95% to achieve the same effective concentration of active compound. This can translate into significant savings over time.
Common Impurities in Peptide Synthesis
Origins of Impurities
Solid-Phase Peptide Synthesis (SPPS) is a complex chemical process in which amino acids are assembled one at a time on a solid support. Each coupling step (addition of an amino acid) has an efficiency below 100%, generating characteristic impurities.
Main Types of Impurities
Deletion peptides: these occur when an amino acid fails to be incorporated into the sequence. The result is a peptide missing one or more residues. For a 39-amino-acid peptide like TRIPLE-G (retatrutide), the deletion of a single residue produces a peptide with a molecular weight approximately 100–200 Da lower.
Truncated peptides: synthesis terminates prematurely, generating fragments of the target sequence. These are typically the major impurities in low-purity peptides.
Oxidized forms: methionine and tryptophan can become oxidized during synthesis or purification. Methionine sulfoxide is the most common oxidation impurity.
Epimers (D-amino acids): during coupling, partial racemization can convert L-amino acids into D-amino acids, generating diastereomers of the target peptide.
Deamidated peptides: asparagine and glutamine can undergo deamidation (conversion to aspartic acid and glutamic acid, respectively), generating impurities with different charge.
Residual coupling agents and solvents: traces of HBTU, HATU, DMF, NMP, or other synthesis reagents may co-purify with the peptide.
Quantification of Impurities
A complete HPLC report identifies not only the total purity but also individual impurities with their relative abundance. A high-quality report includes:
- Main peak purity (%)
- List of detectable impurities with their retention time and abundance (%)
- Method detection limit
- Analytical conditions (column, gradient, detector)
How to Verify Purity Claims
The Problem of Self-Reporting
The research peptide market has a structural problem: many suppliers claim high purities (99%+) without providing verifiable analytical evidence. Purity claims unsupported by data are, at best, unreliable.
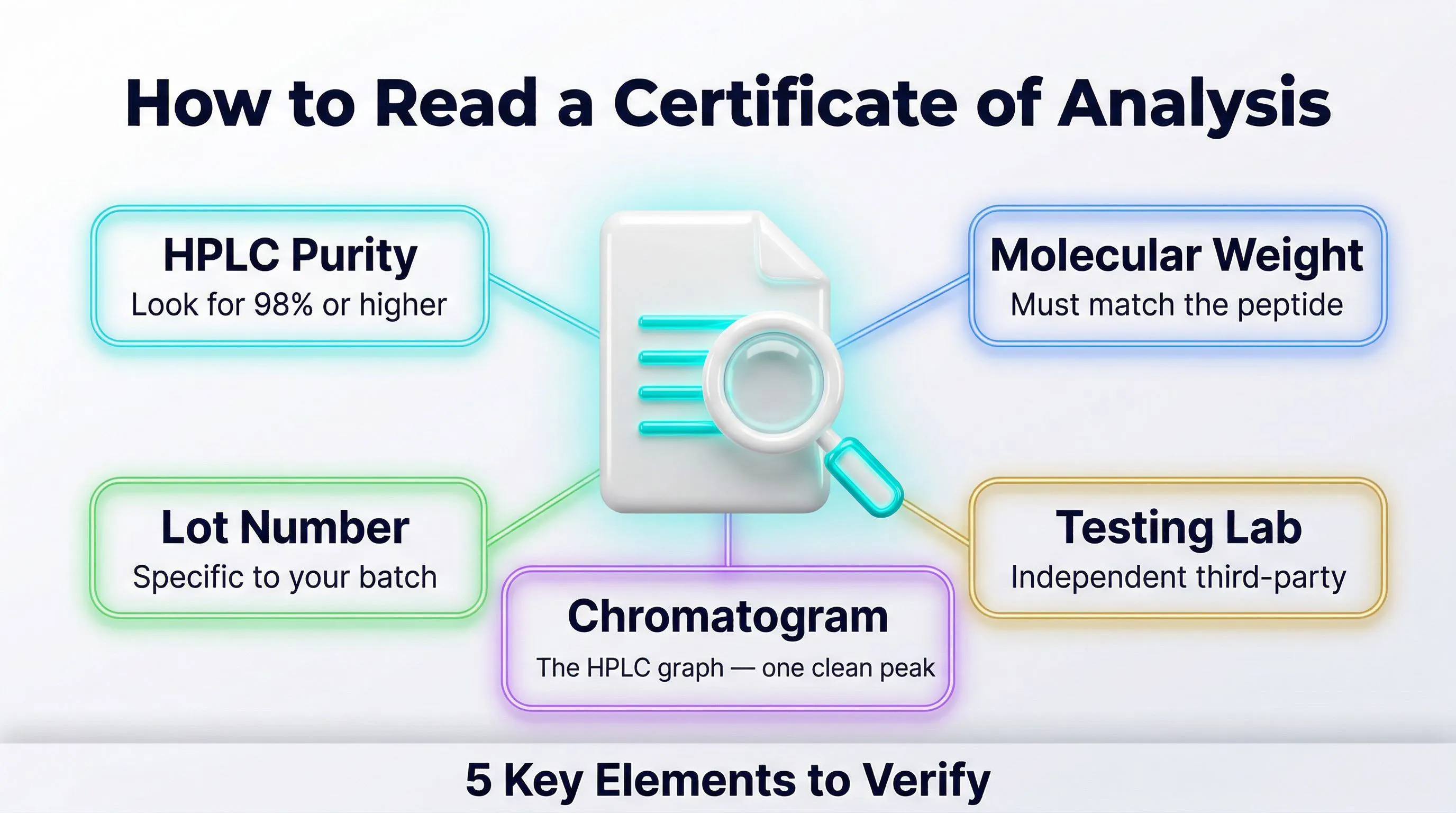
Internal Certificate of Analysis (CoA)
The first level of verification is the Certificate of Analysis (CoA) provided by the manufacturer or supplier. A credible CoA must include:
- Date of analysis
- Peptide lot number
- HPLC method used (column type, gradient, wavelength)
- Complete chromatogram (not just the purity number)
- Mass spectrometry result (measured vs. theoretical molecular weight)
- Name and signature of the analyst or quality manager
Red flags in a CoA:
- Only the purity number without a chromatogram — the value may be fabricated
- Chromatogram with low resolution or an overly short time axis — impurities may be hidden
- No mass data — the peptide’s identity is unconfirmed
- Generic format without a lot number — it could be a reused template
Third-Party Testing: The Gold Standard
The most reliable way to verify peptide purity is independent third-party testing. The most respected laboratory in the research peptide sector is Janoshik Analytical (Czech Republic), which offers:
- HPLC analysis with a complete chromatogram
- Mass spectrometry for identity confirmation
- Independent and unbiased report
- Publishable and verifiable results
A supplier that voluntarily submits its products for Janoshik testing demonstrates confidence in the quality of its peptides. Conversely, a supplier that refuses or discourages independent testing should raise suspicion.
DIY Testing
Researchers with access to an HPLC system can verify the purity of purchased peptides in-house. The standard method involves:
- Preparing a solution of the peptide in water or diluted acetonitrile
- Injecting 5–20 microliters into the HPLC system
- Using a C18 column with a linear water/acetonitrile + 0.1% TFA gradient
- Detecting at 214 nm or 220 nm
- Calculating purity from peak area integration
The Cost-Purity Trade-Off
Why Purity Is Expensive
The price of a peptide increases significantly with the required purity for a straightforward reason: purification has diminishing returns. To obtain a peptide at 99%+ starting from crude (~70%), the following steps are required:
- Preparative HPLC purification (the same analytical technique, but on a larger scale)
- Rigorous fraction selection: only the central fractions of the peak, with maximum purity, are collected
- Side fractions (containing impurities) are discarded — material loss
- To reach 99.5%+, a second purification may be necessary — further loss
The result is that from 100 mg of crude peptide, you might obtain:
| Target Purity | Typical Yield | mg Obtained from 100 mg Crude |
|---|---|---|
| >95% | 50–70% | 50–70 mg |
| >98% | 30–50% | 30–50 mg |
| >99% | 20–35% | 20–35 mg |
| >99.5% | 10–25% | 10–25 mg |
| >99.8% | 5–15% | 5–15 mg |
Material loss during purification is the primary driver of high-purity peptide costs.
When It Is Worth Investing in Purity
The choice of purity grade depends on the application:
>95% is sufficient for:
- Initial screening of peptide libraries
- Qualitative binding studies (yes/no)
- Analytical method development
- Exploratory experiments
>98% is recommended for:
- Most standard research studies
- In vitro cell-based assays
- Quantitative binding studies
- Preliminary biological activity studies
>99% is necessary for:
- Precise dose-response studies
- In vivo experiments
- Pharmacokinetic studies
- Publications in peer-reviewed journals
>99.5% is ideal for:
- Reference standards
- GLP (Good Laboratory Practice) studies
- Analytical method validation
- Research intended to support regulatory filings
For those who want to learn more about how to read a certificate of analysis and compare purity levels, on fornitori europei specializzati you will find detailed guides and Janoshik reports published for every lot, with purities in the ultra-premium range (at or above 99.8%).
Techniques Complementary to HPLC
Mass Spectrometry (MS)
While HPLC measures purity, mass spectrometry confirms the identity of the peptide. The two techniques are complementary:
- HPLC: answers the question “how pure is it?”
- MS: answers the question “is it the right peptide?”
The most widely used MS techniques for peptides are:
- ESI-MS (Electrospray Ionization): the most common, generating multiply charged ions from the peptide
- MALDI-TOF (Matrix-Assisted Laser Desorption/Ionization – Time of Flight): excellent for peptides and proteins
- LC-MS (Liquid Chromatography – Mass Spectrometry): combines HPLC and MS in a single instrument, allowing identification of every peak in the chromatogram
Amino Acid Analysis (AAA)
Amino acid analysis hydrolyzes the peptide into its constituent amino acids and quantifies them individually. This confirms:
- The amino acid composition of the peptide
- The actual peptide content (net of counterion and water)
- The correct amino acid stoichiometry
LAL Endotoxin Testing
For peptides intended for in vivo studies or sensitive cell-based assays, the LAL test (Limulus Amebocyte Lysate) is essential to rule out bacterial endotoxin contamination (lipopolysaccharides). Endotoxins can:
- Activate the innate immune system
- Alter immunological assay results
- Cause fever and septic shock in in vivo studies
Storage and Stability: How to Protect Purity
A peptide purchased at 99.8% purity will not maintain that purity indefinitely unless stored correctly. The main degradation factors are:
Moisture
Lyophilized peptides are hygroscopic and absorb water from the environment. Moisture accelerates degradation reactions (hydrolysis, deamidation). Store lyophilized peptides in airtight containers, ideally with a silica gel desiccant packet.
Temperature
Chemical degradation reactions approximately double in rate for every 10 degrees Celsius increase in temperature. Store peptides at -20 degrees Celsius for medium-term use (months) or at -80 degrees Celsius for long-term storage (years). Avoid repeated freeze-thaw cycles.
Light
UV radiation can degrade photosensitive amino acids (tryptophan, tyrosine). Store peptides in amber vials or protected from light.
Oxidation
Atmospheric oxygen can oxidize methionine, cysteine, and tryptophan. For long-term storage, consider storage under an inert atmosphere (nitrogen or argon).
Reconstituted Peptides
Once reconstituted in solution, peptide stability decreases significantly. General guidelines:
- Aqueous solutions at 4 degrees Celsius: stable for days to weeks
- Aqueous solutions at -20 degrees Celsius: stable for months
- Aliquot to avoid freeze-thaw cycles
- Add 0.1% bovine serum albumin (BSA) to prevent adsorption to containers at low concentrations
Conclusions
HPLC purity is not a technical detail — it is the fundamental parameter that determines the quality of a peptide and, consequently, the validity of the results you obtain with it. The difference between a peptide at 95% and one at 99.8% translates into concrete differences in reproducibility, dosage accuracy, and the absence of artifacts.
This is even more true for complex molecules like TRIPLE-G (retatrutide), with its 39 amino acids: the longer the chain, the more numerous the potential synthesis impurities. When you work with a triple agonist that activates three different receptors (GLP-1, GIP, and glucagon), you want to be certain that what you have in the vial is exactly what you expect.
For those who want to go deeper, investing in the quality of starting materials is always more efficient than trying to figure out why results are not adding up.
References
- Mant CT, Hodges RS. “High-Performance Liquid Chromatography of Peptides and Proteins.” CRC Press. 1991.
- Rabel SR, Stobaugh JF. “Applications of capillary electrophoresis in pharmaceutical analysis.” Pharm Res. 1993.
- ICH Q6B. “Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products.” International Council for Harmonisation, 2023.
- USP <621>. “Chromatography.” United States Pharmacopeia, General Chapter.
- European Pharmacopoeia 11.0. “2.2.29. Liquid Chromatography.” General monograph.
- Coin I, et al. “Solid-phase peptide synthesis: from standard procedures to the synthesis of difficult sequences.” Nat Protoc. 2007;2(12):3247-3256.
- Janoshik Analytical Services. “Peptide Analysis Methodology.” Technical documentation.
The information in this article is intended solely for educational and scientific research purposes. It does not constitute professional advice on product quality. For peptide analysis, always consult the guidelines and protocols of your institution.
Frequently Asked Questions
What is a good HPLC purity level for research peptides?
For most standard research, HPLC purity of 98% or higher is recommended. For precise dose-response studies and in vivo experiments, 99% or higher is necessary. Premium or ultra-premium grade peptides at 99.5% or above are ideal for reference standards and GLP studies. The higher the purity, the fewer confounding impurities in your results.
What is the difference between 95% and 99% purity in a peptide?
The difference is far larger than it appears. A 10 mg vial at 95% purity contains 0.5 mg of impurities, while the same vial at 99% contains only 0.1 mg — five times less. These impurities can have their own biological activity, interfere with assays, or introduce toxicity, so minimizing them directly improves result reliability.
How does HPLC separate peptide impurities from the target compound?
Reversed-phase HPLC separates compounds based on hydrophobicity. The peptide solution is pushed through a column packed with nonpolar material (C18 chains) using a water-to-acetonitrile gradient. More hydrophilic impurities elute first, while more hydrophobic ones elute later, separating the target peptide from synthesis byproducts like truncated sequences, oxidized forms, and deletion peptides.
Why is third-party HPLC testing more reliable than manufacturer testing?
Manufacturer-provided purity data involves an inherent conflict of interest, as the seller benefits from reporting higher numbers. Independent third-party laboratories like Janoshik Analytical test without commercial bias, providing verifiable chromatograms and mass spectrometry data. A supplier that voluntarily submits products for independent testing demonstrates genuine confidence in quality. Learn more in our COA reading guide.
Where can I source research-grade peptides in Europe?
For research requiring high-purity peptides, prioritize suppliers that provide lot-specific HPLC chromatograms and independent third-party testing results. fornitori europei specializzati is a verified European supplier offering HPLC purity of 98% or higher with published Janoshik reports, COA with every lot, free EU shipping, and cryptocurrency payment options.