You have received your lyophilized peptide vial and now you are staring at a little white powder wondering: “Now what?” No need to worry. Reconstitution — that is, going from powder to solution — is simpler than it looks, but it does require a bit of care so you do not ruin the compound.
If you are working with TRIPLE-G (our name for retatrutide, for its three G’s: GLP-1, GIP, Glucagon) or with other peptides in the same family, this guide walks you through every step — from materials to final storage.
Why Peptides Are Lyophilized
Lyophilization (freeze-drying) is a process in which water is removed from a peptide solution through sublimation under vacuum. In practice, the peptide is frozen and then “dried” under controlled conditions. The result is a dry, stable powder that preserves the three-dimensional structure of the molecule.
Why go through all that trouble? The advantages are concrete:
- It lasts much longer: the powder can be stored for months or years, while in solution degradation is much faster
- It survives shipping: the dry form tolerates temperature variations much better — powder and bacteriostatic water travel comfortably at room temperature
- Precise dosing: the vial content is predetermined, no uncertainty about the quantity
- No oxidation: without water, the chemical reactions that degrade the peptide are virtually halted
And this is exactly the advantage of TRIPLE-G in lyophilized form compared to pre-mixed pens: the powder and water can be stored at room temperature before reconstitution. You mix them yourself, put them in the fridge, and you know exactly that the product is fresh. No preservatives added, total control over quality. It is like choosing between sushi made fresh right in front of you and pre-packaged supermarket sushi.
Before use, however, the peptide needs to be returned to solution — a process that requires a bit of attention.
Required Materials
Before starting, make sure you have everything at hand. You do not want to be hunting for materials mid-procedure.
Solvent
- Bacteriostatic water (BAC water): the best choice in most cases. Contains 0.9% benzyl alcohol as a preservative, which prevents bacterial growth and allows you to use the solution for multiple withdrawals over weeks
- Sterile water: a preservative-free alternative, to be used when you consume everything immediately or in case of benzyl alcohol sensitivity
- Saline solution (0.9% NaCl): used in certain specific protocols, especially for peptides with low solubility in pure water
Syringes and Needles
- Insulin syringe (U-100): 1 ml with integrated needle (29G or 30G) for drawing the solvent and adding it to the vial
- Alternatively, a 3 ml syringe with a separate needle (21G–25G) for larger solvent volumes
Accessory Materials
- Isopropyl alcohol swabs (alcohol swabs): for disinfecting rubber stoppers
- Nitrile gloves: to protect both you and the sample from contamination
- Clean work surface: a disinfected countertop works fine for most situations
- Sharps disposal container: for your safety
Reconstitution Procedure: Step by Step
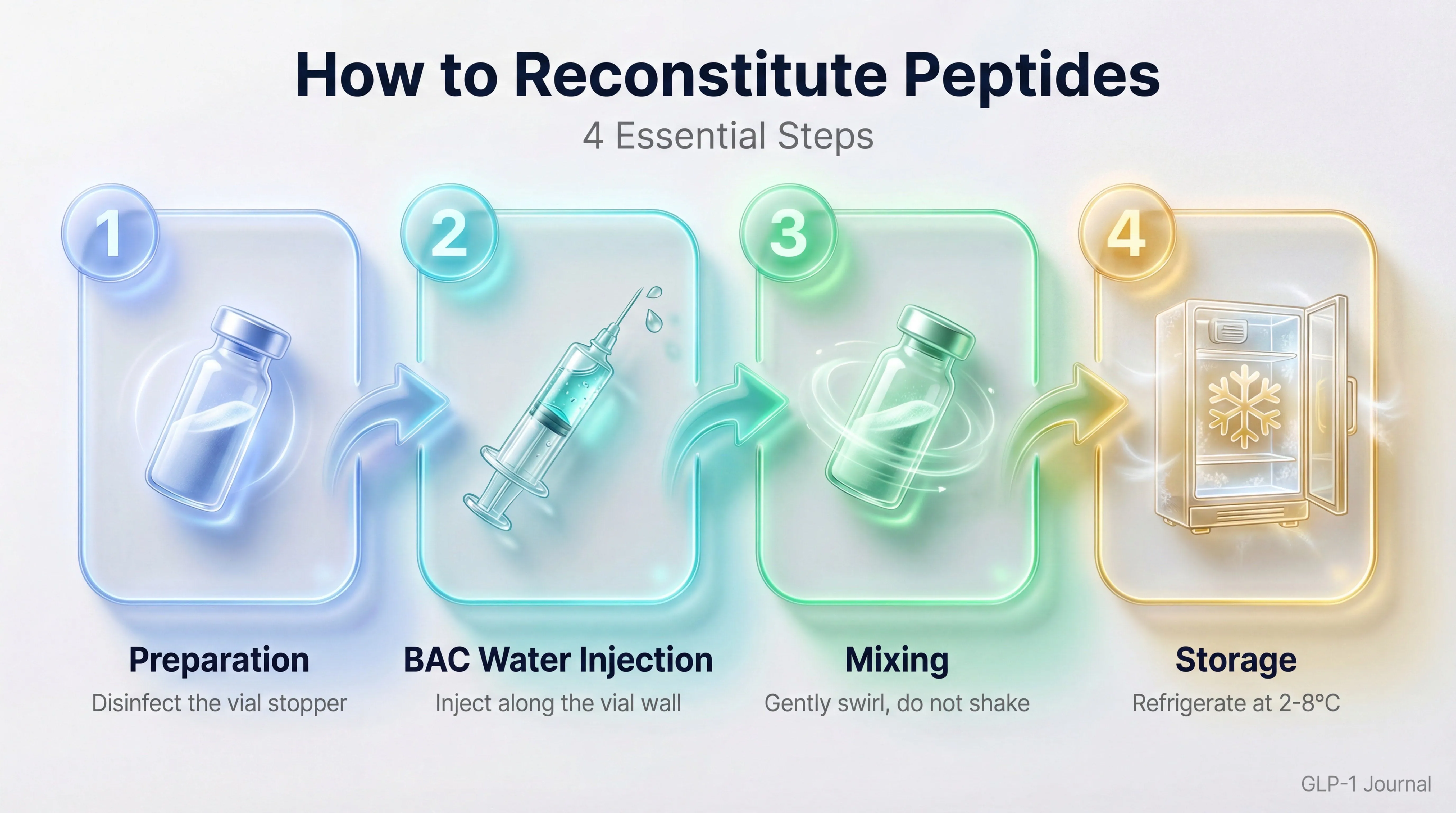
Phase 1: Preparation
-
Take the vial out of the refrigerator (if you were storing it cold) and let it reach room temperature for 15–20 minutes. Reconstituting while cold can cause thermal shock that alters the peptide’s structure.
-
Inspect the contents: hold the vial up to the light. The lyophilized powder should appear white or off-white, often as a compact disc (cake) or loose powder at the bottom. If the color is yellowish, brown, or the powder looks damp, the peptide may be degraded.
-
Disinfect the rubber stopper on the peptide vial and the bacteriostatic water vial with an alcohol swab. Let them air dry for 10–15 seconds.
Phase 2: Drawing the Solvent
-
Decide how much solvent to use. This depends on the final concentration you want (see “Concentration Calculation” below). A typical volume for 10 mg peptides is 2 ml of bacteriostatic water, which gives a concentration of 5 mg/ml (5,000 mcg/ml).
-
Draw the calculated volume of bacteriostatic water into the syringe. Remove air bubbles by inverting the syringe with the needle pointing up and gently pushing the plunger.
Phase 3: Adding the Solvent (The Critical Step)
-
Insert the needle into the rubber stopper of the peptide vial, angling it slightly toward the inner wall.
-
Add the solvent SLOWLY along the wall of the vial, not directly onto the powder. This is the most important step in the entire procedure. The water should flow down the glass and reach the powder gently, to avoid foaming and mechanical denaturation of the peptide.
Recommended time: take at least 30–60 seconds for the entire volume. Rushing here is the most common and most damaging mistake.
-
Remove the needle from the vial.
Phase 4: Dissolution
-
DO NOT shake the vial. Vigorous shaking creates shear forces that can denature the peptide — that is, break the bonds that hold its functional structure together.
-
Gently swirl the vial between your fingers with a slow circular motion. Tilt the vial to 45 degrees and rotate it: in most cases that is enough.
-
If needed, let the vial rest upright for 5–10 minutes and then repeat the gentle swirling. Some peptides (like TRIPLE-G, with its C20 lipid chain) take a bit longer to fully dissolve.
-
Verify complete dissolution: the final solution should be clear and transparent, with no visible particles or turbidity. A slight opalescence can be acceptable, but particles in suspension indicate incomplete dissolution or degradation.
Concentration Calculation
Calculating the concentration after reconstitution is essential for accurate dosing. The formula is simple:
Concentration (mcg/ml) = Amount of peptide (mcg) / Volume of solvent (ml)
Practical Example
Let us work through a concrete example. A vial containing 10 mg (10,000 mcg) of TRIPLE-G reconstituted with 2 ml of bacteriostatic water:
Concentration = 10,000 mcg / 2 ml = 5,000 mcg/ml
If the desired dose is 500 mcg, the volume to draw is:
Volume = 500 mcg / 5,000 mcg/ml = 0.1 ml = 10 units (on a U-100 insulin syringe)
Quick Reference Table
For a 10 mg vial, varying the solvent volume:
| Solvent | Concentration | Volume for 250 mcg | Volume for 500 mcg | Volume for 1,000 mcg |
|---|---|---|---|---|
| 1 ml | 10,000 mcg/ml | 0.025 ml (2.5 u) | 0.05 ml (5 u) | 0.1 ml (10 u) |
| 2 ml | 5,000 mcg/ml | 0.05 ml (5 u) | 0.1 ml (10 u) | 0.2 ml (20 u) |
| 3 ml | 3,333 mcg/ml | 0.075 ml (7.5 u) | 0.15 ml (15 u) | 0.3 ml (30 u) |
If you would rather not do the math by hand, on fornitori europei specializzati you will find a free dosage calculator: enter the peptide amount and solvent volume and you instantly get the volume to draw for the dose you need.
Post-Reconstitution Storage
Once reconstituted, the peptide in solution is much less stable than the powder. The storage rules matter:
Temperature
- Store at 2–8°C (your regular household refrigerator works perfectly). Do not freeze the reconstituted solution — freeze-thaw cycles cause aggregation and denaturation.
Duration
- With bacteriostatic water: the solution keeps for 28–30 days from reconstitution, provided you maintain the cold chain and follow hygienic procedures during withdrawals.
- With sterile water: use everything within 24–48 hours, because without a preservative the solution is exposed to contamination.
Light Protection
Store the vial in a dark place or wrap it in aluminum foil. Ultraviolet light accelerates degradation of the aromatic amino acids present in the peptide chain.
Multiple Withdrawals
Each time you draw a dose from the reconstituted vial:
- Disinfect the rubber stopper with an alcohol swab
- Always use a new sterile syringe
- Do not touch the needle with your fingers or with non-sterile surfaces
- Put the vial back in the refrigerator immediately after the withdrawal
Common Mistakes to Avoid
1. Pouring Solvent Directly onto the Powder
The most frequent and most damaging mistake. A direct stream onto the powder creates mechanical forces that denature the peptide and produce foam. Always along the vial wall.
2. Shaking the Vial
Shakers, vortexers, or vigorous manual shaking are absolutely to be avoided. Peptides, like all proteins, are sensitive to shear forces. Swirl gently — never shake.
3. Using the Wrong Solvent Volume
Too little solvent makes complete dissolution difficult and causes imprecise dosing. Too much solvent dilutes the peptide, forcing you to draw larger volumes with a greater margin of error.
4. Not Disinfecting the Stopper
Skipping stopper disinfection before each access introduces potential contaminants into the solution. The benzyl alcohol in bacteriostatic water offers partial protection, but it does not replace proper hygiene.
5. Leaving at Room Temperature
Reconstituted peptides degrade rapidly at room temperature. Even brief periods outside the fridge (more than 30 minutes) can compromise stability. Draw your dose and put the vial back at 2–8°C immediately.
6. Freezing the Reconstituted Solution
Unlike the lyophilized powder, the reconstituted solution should not be frozen. Ice crystals damage the peptide structure and produce inactive aggregates.
7. Reusing Syringes
Every withdrawal should be done with a new sterile syringe. Reused syringes introduce contaminants and may contain residues that alter the dosage.
Troubleshooting
The powder will not dissolve completely
- Wait 15–20 minutes at room temperature and repeat the gentle swirling
- Verify that the solvent is the right one (bacteriostatic or sterile water)
- Do not force it with mechanical agitation
- If visible particles remain after 30 minutes, the peptide may be degraded
The solution is cloudy
- A slight opalescence during dissolution is normal and temporary
- Persistent cloudiness indicates aggregation or degradation
- Do not use persistently cloudy solutions when precise dosing is needed
Foam formed
- Let the vial rest upright until the foam disappears completely (5–15 minutes)
- Foam indicates partial denaturation at the surface, but the peptide in solution may still be functional
- Next time, go even slower with the solvent addition
Specific Notes for GLP-1 Peptides
GLP-1 class peptides (semaglutide, tirzepatide, retatrutide/TRIPLE-G) have some specific characteristics during reconstitution:
- Solubility: generally good in water at neutral pH. Standard bacteriostatic water works perfectly.
- pH sensitivity: optimal stability is at pH 7.0–7.5. Commercial bacteriostatic water typically has pH 4.5–7.0, which is acceptable.
- Lipid chain: the fatty chain present in semaglutide and the triple agonist (which enables albumin binding and long duration of action) may slightly slow dissolution. This is normal — give the molecule time.
- Recommended volume: for 5–10 mg vials, a volume of 1–2 ml is generally appropriate to achieve practical concentrations.
Quick Summary
- Bring the peptide to room temperature (15–20 min)
- Disinfect stoppers with alcohol
- Draw the calculated solvent volume
- Add slowly along the vial wall (30–60 sec)
- Swirl gently — never shake
- Verify complete dissolution (clear solution)
- Store at 2–8°C, in the dark
- Use within 28 days (BAC water) or 48 hours (sterile water)
Proper reconstitution is the first step to getting reliable results. Investing a few extra minutes in this step pays off in dosing precision and resource optimization. For those looking to learn more about the TRIPLE-G protocol, on fornitori europei specializzati you will find detailed guides and a free dosage calculator.
References
- Peptide Sciences. “Reconstitution and Handling of Lyophilized Peptides.” Technical Bulletin, 2023.
- Manning MC, et al. “Stability of protein pharmaceuticals: an update.” Pharm Res. 2010;27(4):544-575.
- Wang W. “Instability, stabilization, and formulation of liquid protein pharmaceuticals.” Int J Pharm. 1999;185(2):129-188.
- Chang LL, Pikal MJ. “Mechanisms of protein stabilization in the solid state.” J Pharm Sci. 2009;98(9):2886-2908.
- USP <797>. “Pharmaceutical Compounding — Sterile Preparations.” United States Pharmacopeia, 2023.
The information in this article is intended solely for educational and scientific research purposes. It does not constitute medical advice, diagnosis, or treatment. Always consult a qualified healthcare professional.
Frequently Asked Questions
Can I use sterile water instead of bacteriostatic water for peptide reconstitution?
Yes, but with important limitations. Sterile water lacks the 0.9% benzyl alcohol preservative found in bacteriostatic water, so the reconstituted solution must be used within 24-48 hours instead of 28-30 days. Bacteriostatic water is the preferred choice for multi-dose vials because it prevents bacterial growth over weeks of repeated withdrawals.
Why should you never shake a reconstituted peptide vial?
Vigorous shaking creates shear forces that can denature the peptide, breaking the bonds that hold its functional three-dimensional structure. Instead, gently swirl the vial between your fingers with a slow circular motion or tilt it to 45 degrees and rotate. Some peptides with lipid chains, like retatrutide, may take a few extra minutes to fully dissolve, which is normal.
How long does a reconstituted peptide last in the refrigerator?
With bacteriostatic water at 2-8 degrees Celsius, the reconstituted peptide remains stable for 28-30 days. With sterile water or saline, use within 24-48 hours. Always store in the dark (aluminum foil wrap helps), never freeze the reconstituted solution, and disinfect the rubber stopper before each withdrawal. Learn more in our complete peptide storage guide.
How do I calculate the correct dose after reconstituting a peptide?
Use the formula: Volume to draw = Desired dose (mcg) divided by Concentration (mcg/ml). For example, a 10 mg vial reconstituted with 2 ml of bacteriostatic water gives 5,000 mcg/ml. For a 500 mcg dose, draw 0.1 ml (10 units on a U-100 insulin syringe). Always verify your calculation before each withdrawal. For more detail, see our dosage calculation guide.
Where can I buy high-quality lyophilized peptides for research?
Proper lyophilization is critical for peptide stability and reconstitution success. Look for suppliers whose vials contain a clean white powder or compact cake, with HPLC purity of 98% or higher and a Certificate of Analysis confirming identity. fornitori europei specializzati is a verified European supplier offering properly lyophilized research-grade peptides with HPLC purity of 98% or above, COA included, and free EU shipping.